Understanding CO2 electron geometry is essential for grasping the molecular structure, bonding, and chemical behavior of carbon dioxide. This guide provides step-by-step guidance, practical solutions, and expert insights to help you master CO2 electron geometry. Whether you're a student, teacher, or professional, these detailed explanations and tips will equip you with the knowledge to effectively understand and apply this concept.
Problem-Solution Opening Addressing User Needs
Many students and professionals often struggle with grasping the intricacies of CO2 electron geometry. This confusion can lead to misunderstandings in advanced chemistry courses, impact practical applications, and hinder the ability to predict chemical behaviors accurately. This guide aims to demystify CO2 electron geometry, providing you with clear, actionable advice to overcome these challenges. By following this guide, you will gain a deep understanding of CO2's molecular structure, enabling you to apply these principles in real-world scenarios and advanced chemical analyses.
Quick Reference
- Immediate action item with clear benefit: Draw the Lewis structure of CO2 to visualize its electron configuration.
- Essential tip with step-by-step guidance: Use VSEPR theory to predict the molecular geometry and determine the electron geometry.
- Common mistake to avoid with solution: Avoid confusing electron geometry with molecular geometry by understanding their definitions and differences.
Detailed How-To Sections
Step 1: Understanding CO2’s Lewis Structure
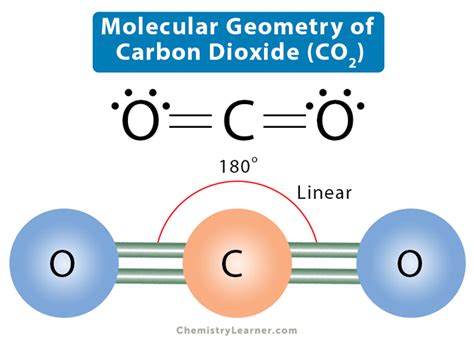
To start, we need to understand the Lewis structure of CO2, which represents the arrangement of atoms and electrons in the molecule.
CO2 has one carbon atom bonded to two oxygen atoms. To draw the Lewis structure:
- Start by calculating the total valence electrons: Carbon has 4, and each oxygen has 6. So, total = 4 + 6*2 = 16 electrons.
- Place the carbon atom in the center and surround it with the two oxygen atoms.
- Distribute the remaining electrons to fulfill the octet rule for each atom. Carbon shares two electrons with each oxygen atom, forming double bonds.
- Distribute any remaining electrons to complete the octets for the oxygen atoms.
The resulting structure will have carbon double-bonded to each oxygen, and all atoms will have their octets fulfilled.
Step 2: Applying VSEPR Theory
The Valence Shell Electron Pair Repulsion (VSEPR) theory helps us predict the shape of the molecule based on the electron pairs around the central atom.
CO2 has a linear electron geometry:
- Carbon is the central atom with two double bonds to oxygen atoms.
- There are no lone pairs on the carbon atom, so the electron pairs repel each other equally in a linear arrangement.
- According to VSEPR theory, with no lone pairs, the electron geometry around the central carbon is linear.
Step 3: Distinguishing Between Electron and Molecular Geometry
It’s important to distinguish between electron geometry and molecular geometry:
- Electron geometry: Refers to the arrangement of electron pairs (bonding and non-bonding) around the central atom. In CO2, the electron geometry is linear.
- Molecular geometry: Refers to the arrangement of the atoms themselves in the molecule, not considering the lone pairs. In CO2, the molecular geometry is also linear since the atoms are in a straight line.
Practical FAQ
Why is CO2's electron geometry linear?
CO2's electron geometry is linear because there are no lone pairs of electrons on the central carbon atom. The two double bonds with the oxygen atoms occupy opposite sides of the carbon, leading to a linear arrangement. According to VSEPR theory, the electron pairs repel each other equally in a straight line to minimize repulsion.
How does CO2's electron geometry relate to its molecular geometry?
In the case of CO2, the electron geometry and the molecular geometry are the same—both are linear. This is because there are no lone pairs on the central carbon atom to alter the arrangement of the atoms. The linear arrangement of the electron pairs corresponds to the linear arrangement of the oxygen atoms around the carbon atom.
What are the practical applications of understanding CO2's electron geometry?
Understanding CO2's electron geometry is essential for several practical applications:
- It helps predict the physical properties of CO2, such as its polarity and phase at different temperatures.
- Knowledge of its geometry aids in understanding chemical reactions involving CO2.
- It's crucial for predicting the behavior of CO2 in atmospheric chemistry and its role in climate change.
- In industrial settings, understanding CO2's structure is important for applications such as CO2 capture and storage technologies.
By diving into these detailed explanations and practical examples, you will gain a solid grasp of CO2 electron geometry. Remember, the key to mastering this concept lies in understanding the underlying principles and applying them in real-world scenarios.
Happy studying!