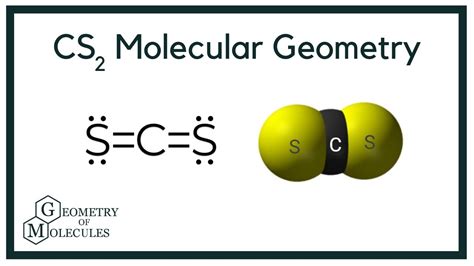Unraveling CS2 Molecular Geometry: A Deep Dive into Its Structure
Are you curious about the intricacies of CS2’s molecular geometry? Whether you’re a chemistry student, a researcher, or simply someone interested in molecular structures, this guide will demystify the topic with step-by-step guidance, real-world examples, and actionable advice. We’ll tackle the fundamental concepts to provide you with a comprehensive understanding of how CS2 is structured at a molecular level.
Introduction to CS2 Molecular Geometry
Carbon disulfide (CS2) is a fascinating molecule often discussed in advanced chemistry courses due to its simple yet intriguing structure. CS2 comprises a carbon atom double-bonded to two sulfur atoms. Understanding its molecular geometry provides insight into its chemical behavior and reactivity.To start, let’s break down the problem-solution approach to address the specific needs of learners and professionals seeking to grasp the CS2 molecular geometry:
Many users often struggle with understanding molecular geometry because they lack a clear, step-by-step explanation combined with practical examples. This guide aims to resolve that by providing thorough explanations, practical examples, and focusing on actionable advice to foster a deeper understanding.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Draw Lewis structures to determine the electron geometry first.
- Essential tip with step-by-step guidance: Use the VSEPR theory to predict molecular shape.
- Common mistake to avoid with solution: Confusing electron geometry with molecular geometry.
Detailed Understanding of Molecular Geometry
To unravel the molecular geometry of CS2, we need to start with electron geometry and then apply the Valence Shell Electron Pair Repulsion (VSEPR) theory. Here’s a detailed guide to understand these concepts:
Electron Geometry
Electron geometry considers all electron pairs around the central atom. These include bonding pairs and lone pairs. For CS2:
1. Count the number of valence electrons:
Carbon (C) has 4 valence electrons, and each sulfur (S) has 6 valence electrons. So, CS2 has a total of 8 valence electrons.
2. Distribute electrons in pairs:
Each double bond counts as 2 electron pairs. Here’s a Lewis structure for CS2:
C = S - S
We place 2 electrons between C and each S to form double bonds, using up 4 electrons. We have 4 electrons left, which will complete the octets for sulfur atoms.
This results in an electron geometry of linear because all electrons are involved in bonding pairs.
Molecular Geometry
Unlike electron geometry, molecular geometry only considers the atoms bonded to the central atom. Applying VSEPR theory, we see that:
1. Central atom (Carbon) has two bonded atoms (each sulfur atom).
2. There are no lone pairs on the carbon atom.
According to VSEPR theory, two atoms bonded to the central atom and no lone pairs result in a linear molecular geometry.
Thus, CS2 has a linear molecular geometry.
Why This Matters
Understanding the geometry of CS2 is crucial for grasping its chemical properties and behavior in various reactions. Knowing the molecular structure aids in predicting the molecule’s polarity, reactivity, and physical properties.
Tips and Best Practices
When studying molecular geometry, here are a few tips to keep in mind:
- Use Visualization Tools: Sketch the molecules to visualize geometry.
- Employ VSEPR Theory: Apply VSEPR to predict the molecular shape accurately.
- Practice Drawing Lewis Structures: Regular practice helps in predicting geometries effectively.
Common Mistakes to Avoid
While studying CS2’s geometry, some common pitfalls can occur:
- Forgetting Lone Pairs: Always check for lone pairs on the central atom as they greatly affect the molecular geometry.
- Overlooking Electron Pairs: Don’t ignore single bonds when counting electron pairs for electron geometry.
- Misinterpreting Geometry: Remember electron geometry is not the same as molecular geometry.
Practical Examples
Here are practical applications and examples to help solidify your understanding of CS2’s molecular geometry:
Example 1: Predicting Geometry Using VSEPR
Predict the molecular geometry of CF2Cl2 (Dichlorodifluoromethane).
1. Determine the Lewis structure:
CF2Cl2 consists of one carbon atom bonded to two fluorine atoms and two chlorine atoms.
2. Count the valence electrons:
Carbon (4), Fluorine (7 each * 2 = 14), Chlorine (7 each * 2 = 14), total = 35 valence electrons.
3. Draw the Lewis structure:
Cl | F - C - F | Cl
4. Apply VSEPR theory:
Carbon has 4 bonded atoms (two fluorines and two chlorines), and no lone pairs. Thus, molecular geometry is tetrahedral.
Practical FAQ
What is the difference between electron geometry and molecular geometry?
Electron geometry considers all electron pairs around the central atom, including both bonding and lone pairs. Molecular geometry, however, only considers the positions of the atoms bonded to the central atom, excluding lone pairs.
For example, in CS2, the electron geometry is linear because it only has bonding pairs, while the molecular geometry remains linear since there are no lone pairs to distort the shape.
How can I visualize molecular geometry?
To visualize molecular geometry, use molecular modeling kits or online molecular modeling software. Draw the Lewis structure first to understand the electron distribution, and then apply VSEPR to predict the shape.
You can also use ball-and-stick models to physically represent atoms and bonds, helping in better spatial comprehension.
Why is CS2 linear?
CS2 is linear because it has two sulfur atoms bonded to a central carbon atom with no lone pairs on the central carbon atom. In a linear molecule, the atoms are arranged in a straight line, maintaining this structure due to the repulsion between bonding pairs which arrange themselves to minimize repulsion.
The VSEPR theory supports this by predicting a linear arrangement given there are two bonding pairs and no lone pairs on the central atom.
This guide offers a comprehensive approach to understanding CS2’s molecular geometry, providing you with practical tools and actionable advice to master this topic.