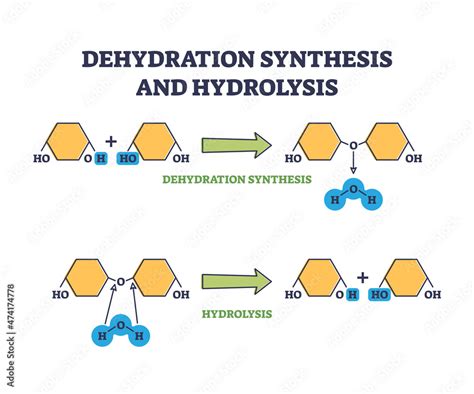
The fundamental aspects of biochemistry involve an intricate dance between chemical reactions. Among these, dehydration synthesis and hydrolysis play pivotal roles. While they both deal with the transformation of molecules, they proceed in opposite directions. Understanding these processes can illuminate critical aspects of biological systems, from metabolic pathways to molecular bonding.
What Is Dehydration Synthesis?
Dehydration synthesis, also known as condensation reactions, involve the joining of two molecules with the concomitant removal of a water molecule. This process is fundamental in forming larger biomolecules from smaller units. For instance, when glucose molecules combine to form cellulose, a water molecule is expelled in the process. This reaction is indispensable in the biosynthesis of carbohydrates, proteins, and nucleic acids, forming complex biological macromolecules from monomers.
Key Insights
- Primary insight with practical relevance: Dehydration synthesis is crucial for creating complex biomolecules from simpler ones, playing a significant role in biological macromolecule formation.
- Technical consideration with clear application: In polymer chemistry, understanding dehydration synthesis aids in synthesizing artificial polymers with applications in medical and industrial sectors.
- Actionable recommendation: Researchers should focus on optimizing conditions for dehydration synthesis to enhance efficiency in macromolecule production.
What Is Hydrolysis?
Conversely, hydrolysis involves breaking down complex molecules into simpler units with the addition of water. This process is pivotal in metabolic pathways where stored molecules are broken down for energy or to reclaim essential building blocks. For example, the hydrolysis of a disaccharide like sucrose breaks it into glucose and fructose, which can then be utilized for energy or other cellular processes. This process is integral to various metabolic pathways and the daily functioning of cells.
Comparative Analysis: Dehydration Synthesis vs. Hydrolysis
While dehydration synthesis and hydrolysis are opposite in nature, both are essential for maintaining the dynamic equilibrium of biological systems. Dehydration synthesis constructs complex structures, which are then broken down via hydrolysis to recycle components and free up energy. These two processes ensure that biological systems can grow, repair, and function effectively. For example, the formation and subsequent breakdown of peptide bonds in proteins via these reactions demonstrate the cyclic nature of molecular interactions in living organisms.
What are some real-life examples of dehydration synthesis?
Dehydration synthesis is involved in the formation of DNA, where nucleotides join to create the long, complex strands of genetic material. Similarly, in the formation of proteins, amino acids undergo dehydration synthesis to create polypeptide chains.
How do enzymes influence dehydration synthesis and hydrolysis?
Enzymes act as catalysts to speed up these reactions. In dehydration synthesis, enzymes lower the activation energy required to form new bonds, while in hydrolysis, they make it easier to break down existing bonds, thus regulating these processes efficiently within biological systems.
Understanding dehydration synthesis and hydrolysis provides insights into how cells build and maintain vital structures and functions. These processes not only explain the fundamental operations of biochemistry but also pave the way for advancements in fields like medicine, biotechnology, and synthetic biology.