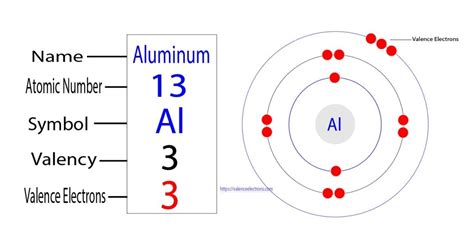
Understanding the electron count in aluminum is crucial for chemists and physicists alike, as it provides fundamental insight into the atom’s structure and behavior. Aluminum, symbolized as Al, is the thirteenth element in the periodic table, and its atomic number is 13. Consequently, a neutral aluminum atom contains 13 electrons. However, exploring deeper into the atomic structure reveals fascinating insights into the electron distribution and configurations that make aluminum unique. This detailed analysis explores the electron count and configuration in aluminum, along with the practical implications in various applications.
Electron Configuration: Unveiling Aluminum’s Secrets
To understand aluminum’s electron count, one must delve into its electron configuration. An electron configuration explains how electrons are distributed in orbitals around the nucleus of an atom. For aluminum, this configuration is written as 1s² 2s² 2p⁶ 3s² 3p¹. This notation reveals that there are two electrons in the first energy level (1s), eight in the second (2s and 2p combined), and three in the third (3s and 3p combined). This specific configuration leads to aluminum’s metallic properties, influencing its role in industry and various chemical reactions.Importance of Electron Count in Aluminum
The electron count in aluminum is pivotal for various practical applications. Knowing that aluminum has 13 electrons allows scientists and engineers to predict its chemical reactivity and behavior in different environments. This knowledge is critical in the fields of materials science and chemistry. For instance, aluminum’s outermost electrons, particularly those in the 3s and 3p orbitals, are easily lost to form a positive ion, which explains aluminum’s reactivity with acids and its prevalence in forming alloys.Key Insights
- Aluminum's electron count directly influences its reactivity and chemical behavior.
- Understanding the electron configuration helps in predicting its metallic properties and applications.
- Knowledge of aluminum's electron structure aids in the development of advanced materials and technologies.
Implications in Industry and Technology
The electron count and configuration of aluminum have significant implications in the tech and industrial sectors. Aluminum’s electron configuration contributes to its low density, high thermal and electrical conductivity, and corrosion resistance, making it an invaluable material in aerospace, automotive, and electronics industries. These properties are harnessed in the production of lightweight components, efficient electrical wiring, and robust, durable consumer products.FAQ Section
What role does the electron count play in aluminum’s conductivity?
The outermost electrons in aluminum are loosely bound, which facilitates the flow of electric current, resulting in high electrical conductivity. This property makes aluminum a preferred material in electrical wiring and components.
How does the electron configuration of aluminum influence its reactivity?
Aluminum's ability to easily lose its three valence electrons (from the 3s and 3p orbitals) makes it highly reactive. This characteristic is fundamental in its corrosion processes and in the creation of aluminum compounds used in various industrial applications.
Understanding the electron count in aluminum not only unravels a fundamental atomic mystery but also opens doors to countless innovations and technological advancements. The practical insights gleaned from this knowledge continue to drive progress across multiple scientific and engineering domains. With these foundational principles, researchers and industry professionals can further develop and utilize aluminum in ways that benefit numerous industries and everyday applications.