Understanding whether a compound like CF4 (carbon tetrafluoride) is polar or nonpolar involves diving into its molecular geometry and the principles of molecular polarity. CF4, an industrially significant compound, serves as an excellent case study for grasping the concepts of molecular geometry and dipole moments. Here, we will delve into its structure to determine whether it is polar or nonpolar, ensuring you understand the intricacies involved in this chemical investigation.
Key insights box:
Key Insights
- CF4 exhibits a nonpolar molecular nature due to its symmetrical tetrahedral geometry.
- The distribution of electron density around the central carbon atom is even, mitigating dipole moments.
- To verify the nonpolarity, calculate and evaluate the net dipole moment.
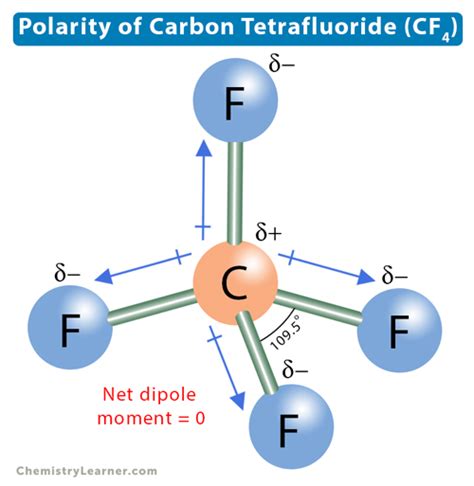
CF4, with its four fluorine atoms symmetrically bonded to a central carbon atom, presents a unique molecular geometry. This geometry forms a regular tetrahedron, where each fluorine atom is equally spaced from the central carbon atom. The symmetrical distribution of the fluorine atoms means that any individual dipole created by the difference in electronegativity between carbon and fluorine atoms is canceled out by its opposite across the molecule.
In a molecule like CF4, the carbon atom, having a lower electronegativity compared to fluorine, forms polar bonds with each fluorine atom. Despite these polar bonds, the symmetrical tetrahedral arrangement leads to a molecule where the dipole moments cancel each other out. This results in a net dipole moment of zero, making the molecule nonpolar.
The second aspect of CF4’s nonpolar nature comes from its molecular orbital theory. In tetrahedral geometry, the symmetric electron cloud around the carbon atom ensures an even distribution of charge, thereby preventing any partial charges from arising. This molecular stability further reinforces the nonpolarity of CF4.
CF4’s practical applications in various industrial processes highlight its stability and nonpolar properties. Its use in the semiconductor industry as a fluorinating agent underscores the molecule’s stability under different conditions, further supported by its lack of polarity which allows it to interact uniformly with other nonpolar substances.
CF4’s unique molecular configuration exemplifies how geometry plays a critical role in determining the polarity of a molecule. Despite the individual bond polarities between carbon and fluorine, CF4’s overall molecular structure negates any net dipole moment, affirming its nonpolar nature.
FAQ section:
What makes CF4 nonpolar despite having polar bonds?
CF4 is nonpolar due to its symmetrical tetrahedral geometry. The equal spacing and arrangement of the four fluorine atoms around the central carbon atom cause the individual bond dipoles to cancel each other out, resulting in a net dipole moment of zero.
How does CF4’s nonpolarity influence its industrial applications?
CF4’s nonpolarity allows it to interact uniformly with nonpolar substances, making it an excellent choice in applications such as semiconductor manufacturing where uniformity and stability are crucial.
In conclusion, CF4’s molecular geometry and symmetry underscore its nonpolar nature despite the individual bond polarities. This unique characteristic makes it an invaluable compound in various industrial applications, particularly where chemical stability and nonreactivity with nonpolar substances are essential.