Is CH3Cl Polar or Nonpolar: Exploring Its Molecular Chemistry
When it comes to understanding molecular chemistry, determining whether a molecule is polar or nonpolar is a key step. This knowledge is not only fundamental for chemists but also incredibly valuable for students and anyone interested in science. Today, we're diving into CH3Cl, or methyl chloride. You might wonder, "Is CH3Cl polar or nonpolar?" Let’s break down this molecule and its properties step-by-step, providing you with practical insights and actionable advice to grasp these concepts easily.
Understanding the polarity of a molecule like CH3Cl isn't just an academic exercise; it has real-world implications in fields ranging from pharmaceuticals to environmental science. Whether you are a student, an industry professional, or just someone curious about chemistry, this guide will help you navigate through the nuances of molecular polarity.
Problem-Solution Opening Addressing User Needs
If you’re reading this guide, you’re likely trying to determine the polarity of CH3Cl and understand what this means for its chemical behavior. You might be facing challenges in grasping these concepts from textbooks or online resources. Fear not! This guide offers a straightforward and comprehensive exploration, breaking down the complexities of molecular chemistry into simple, digestible pieces. By the end of this guide, you'll confidently determine the polarity of CH3Cl, understand its implications, and even apply this knowledge to practical scenarios in chemistry and beyond.
Quick Reference
- Immediate action item: Check the electronegativity difference between the atoms involved.
- Essential tip: Use the VSEPR theory to predict molecular geometry.
- Common mistake to avoid: Forget that a symmetrical molecule can still be polar if it has polar bonds.
Understanding Polarity
Polarity in molecules is a fundamental concept that describes how electrons are shared between atoms and how this affects the overall molecule's properties. A molecule is considered polar if it has a net dipole moment, meaning there is an uneven distribution of electrical charge. Conversely, nonpolar molecules have a balanced distribution of charge.
Key Elements in Determining Polarity
To determine if CH3Cl is polar or nonpolar, we need to delve into several factors:
- Electronegativity: Electronegativity is the ability of an atom to attract electrons in a chemical bond. The difference in electronegativity between atoms in a bond can determine the bond’s polarity.
- Molecular Geometry: The shape of the molecule plays a crucial role in determining polarity. This includes understanding the arrangement of atoms around the central atom.
- Symmetry: Even if a molecule has polar bonds, if it is symmetrical, the dipole moments can cancel each other out, making the molecule nonpolar.
Electronegativity of CH3Cl
First, let's look at the electronegativity of the atoms involved in CH3Cl. Chlorine (Cl) is more electronegative than carbon (C) and hydrogen (H). This difference means that chlorine pulls electrons more strongly than carbon and hydrogen.
Electronegativity values:
- Carbon (C): 2.55
- Hydrogen (H): 2.20
- Chlorine (Cl): 3.16
The significant difference in electronegativity between carbon and chlorine leads to a polar C–Cl bond.
Molecular Geometry of CH3Cl
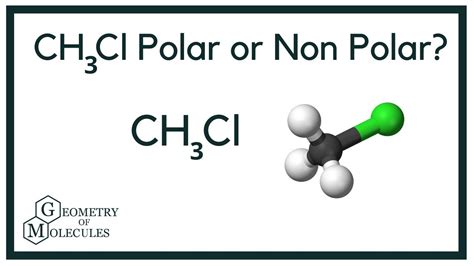
Next, we must consider the molecular geometry. CH3Cl has a tetrahedral molecular geometry. This means that the chlorine atom is bonded to the carbon atom, and the carbon atom is bonded to three hydrogen atoms, forming a pyramid-like shape.
VSEPR Theory (Valence Shell Electron Pair Repulsion) helps us predict the shape of the molecule. In a tetrahedral arrangement, there are four electron pairs around the central carbon atom: three bonding pairs with hydrogen and one with chlorine.
Symmetry and Polarity
A common pitfall is assuming that a tetrahedral shape means the molecule is nonpolar. Even with a symmetrical arrangement, if the individual bonds are polar (as in the C–Cl bond), the molecule can still have a net dipole moment.
In CH3Cl, although the tetrahedral geometry would suggest symmetry, the polar C–Cl bond breaks this symmetry, leading to an overall polar molecule. Therefore, CH3Cl is a polar molecule.
Practical Examples
To make this concept more tangible, let’s look at some practical examples of CH3Cl:
- In industrial applications: CH3Cl is used in the synthesis of various chemicals and pharmaceuticals.
- In environmental science: Understanding the polarity of CH3Cl can help in assessing its solubility and interactions with other environmental molecules.
- In everyday chemistry: Knowing the polarity of a molecule helps predict its behavior in solvents and reactions.
FAQs about CH3Cl Polarity
Why is CH3Cl more soluble in polar solvents than in nonpolar solvents?
CH3Cl, being a polar molecule due to the presence of the polar C–Cl bond, interacts favorably with other polar molecules. According to the principle “like dissolves like,” polar solvents such as water or methanol are more effective at dissolving CH3Cl than nonpolar solvents like hexane. The positive end of the polar solvent molecules surrounds the negative ends of the CH3Cl, and vice versa, creating a solvation shell that allows for effective dissolution.
Can the polarity of CH3Cl affect its boiling and melting points?
Yes, the polarity of CH3Cl affects its boiling and melting points. Polar molecules like CH3Cl experience stronger intermolecular forces known as dipole-dipole interactions compared to nonpolar molecules of similar size and mass. These stronger interactions require more energy to overcome, resulting in higher boiling and melting points for polar molecules. Thus, CH3Cl has higher boiling and melting points compared to nonpolar alkanes with similar molecular weights.
Best Practices and Tips
Here are some best practices and tips to help you understand and apply the concept of molecular polarity more effectively:
- Visualize Molecular Shapes: Use molecular models or software to visualize the molecular geometry and identify potential polarities.
- Calculate Electronegativity Differences: Always calculate the electronegativity difference between atoms in a bond to determine the bond polarity.
- Analyze Symmetry: Even if a molecule has polar bonds, consider its overall symmetry. A symmetrical arrangement can cancel out dipole moments, but a slight asymmetry can create a net dipole.
- Understand Practical Implications: Grasp how polarity affects solubility, boiling and melting points, and interactions with other molecules in various scientific applications.
In conclusion, understanding whether CH3Cl is polar or nonpolar involves comprehending electronegativity, molecular geometry, and symmetry. With this knowledge, you can predict the molecule’s behavior in various chemical contexts. Remember, the key to mastering molecular chemistry lies in breaking down complex ideas into manageable parts and applying practical examples and tips. Whether you are studying for an exam or exploring a new field of science, this guide equips you with the knowledge and tools to understand the polarity of CH3Cl and similar molecules effectively.