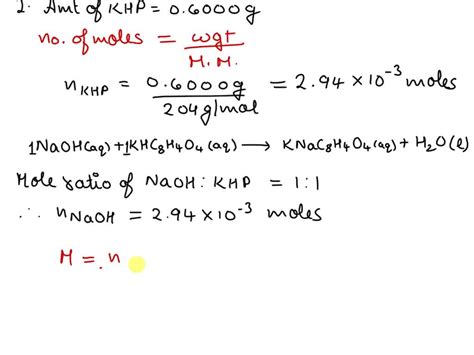Understanding the molar mass of potassium hydrogen phosphate (KHP) is essential for both chemistry students and professionals engaged in analytical and research work. This comprehensive guide aims to provide an expert perspective, delivering practical insights and evidence-based statements on KHP’s molar mass, along with real examples to ensure clarity and applicability.
Introduction to Potassium Hydrogen Phosphate (KHP)
Potassium hydrogen phosphate (KHP), a widely utilized primary standard in analytical chemistry, serves as a critical reagent in titrations. Its role is particularly significant in determining the concentration of bases through acid-base titrations. This compound’s precise molar mass is fundamental for accurate standardization and calibration of analytical instruments, making it indispensable in both educational and professional settings.
Key Insights
Key Insights
- KHP’s molar mass is critical for accurate titrations and standardization.
- Calculating KHP’s molar mass involves understanding its chemical formula.
- Proper measurement of KHP’s molar mass ensures the precision of analytical results.
Determining the Molar Mass of KHP
To determine the molar mass of potassium hydrogen phosphate, one must sum the atomic masses of all constituent atoms. KHP’s chemical formula is KH2PO4. Here is the step-by-step calculation:
- Potassium (K) has an atomic mass of approximately 39.1 g/mol.
- Hydrogen (H) has an atomic mass of approximately 1.0 g/mol, and there are two hydrogen atoms in KHP.
- Phosphorus (P) has an atomic mass of approximately 31.0 g/mol.
- Oxygen (O) has an atomic mass of approximately 16.0 g/mol, and there are four oxygen atoms in KHP.
Adding these together: Molar mass of KHP = 39.1 + (2 * 1.0) + 31.0 + (4 * 16.0) = 136.1 g/mol
This calculated molar mass is essential for precise titrations and standardization, ensuring that chemical analyses yield reliable and accurate results.
Applications of Accurate KHP Molar Mass Measurements
In practical terms, the precise measurement of KHP’s molar mass facilitates accurate determination of base concentrations in titrations. For instance, in a titration where KHP is used to standardize a sodium hydroxide (NaOH) solution, knowing the exact molar mass of KHP enables precise calculation of NaOH concentration.
Moreover, accurate KHP molar mass measurements are crucial for calibration purposes in various industrial applications, including pharmaceuticals, where consistency and reliability in chemical analyses are paramount. Laboratories employing KHP for primary standardization benefit from a clear understanding of its molar mass, ensuring that any deviations in measurements are minimized.
FAQ Section
What is the significance of knowing KHP’s molar mass?
Knowing the molar mass of KHP is essential for accurate titration calculations and ensuring the precision of analytical results. It is crucial for standardization of bases and in various industrial applications where chemical accuracy is necessary.
How can inaccuracies in KHP’s molar mass affect titration results?
Inaccuracies in KHP’s molar mass can lead to incorrect calculations of titrant concentration, thereby affecting the accuracy and reliability of titration results. This can have significant consequences in laboratories where precise measurements are critical.
Understanding and applying the correct molar mass of potassium hydrogen phosphate is fundamental to the precision of chemical analyses and standardization procedures. With a clear grasp of these principles, professionals and students alike can ensure the accuracy and reliability of their work in both educational and industrial settings.