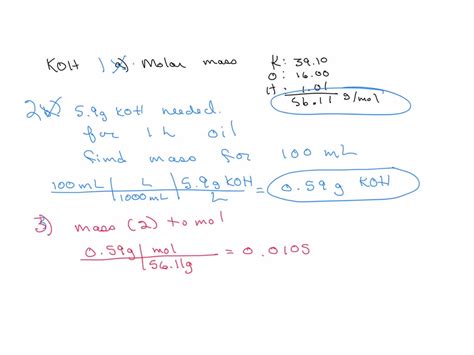The precise determination of molar mass is foundational in chemistry, especially in understanding and predicting chemical reactions and properties. Potassium hydroxide (KOH) serves as a quintessential compound for this examination. With the chemical formula KOH, it consists of a potassium cation (K⁺), an oxygen atom (O), and a hydroxide anion (OH⁻). To decode the molar mass of KOH with precision, we must delve into the atomic weights of its constituent elements. Potassium has an atomic mass of approximately 39.10 g/mol, oxygen weighs around 16.00 g/mol, and hydrogen in the hydroxide group adds about 1.01 g/mol.
Key Insights
- Primary insight with practical relevance: Knowing the molar mass of KOH is crucial for stoichiometric calculations in chemical reactions.
- Technical consideration with clear application: The molar mass of KOH enables precise dilutions and formulations in industrial and laboratory applications.
- Actionable recommendation: Always use up-to-date atomic weight data to calculate the molar mass for accurate experimental results.
Understanding Atomic Weights
The molecular weight or molar mass of a compound is essentially a sum of the atomic weights of all the atoms present in its formula. This requires a precise knowledge of the atomic mass units (amu) for each element in KOH. Potassium (K), as an alkali metal, has an atomic mass of 39.10 amu. Oxygen (O), a non-metal critical in many chemical processes, has an atomic mass of approximately 16.00 amu. The hydrogen (H) component within the hydroxide ion (OH⁻) weighs just 1.01 amu. When combined, the sum is 39.10 + 16.00 + 1.01, yielding a molar mass of 56.11 g/mol for KOH.Applications in Chemical Reactions
Determining the molar mass of KOH provides critical information for executing various chemical processes. In industrial applications, such as the manufacturing of soap and paper, precise calculations using molar mass ensure the correct proportioning of reactants. Within the lab, the molar mass of KOH is indispensable for preparing standard solutions, which are used for calibration and titrations. For instance, when synthesizing a solution of KOH for a titration against an acid, knowing its molar mass allows chemists to calculate the exact quantity needed to achieve a desired concentration.Why is it important to use the most recent atomic weight data?
The atomic mass of elements is subject to periodic revisions based on new research findings. Using outdated atomic weights can lead to inaccurate molar mass calculations, which in turn can affect experimental outcomes and scientific conclusions.
How does molar mass assist in stoichiometry?
In stoichiometry, the molar mass acts as a conversion factor between mass and moles, allowing chemists to determine the quantitative relationships between reactants and products in a chemical reaction.
The detailed and accurate calculation of KOH’s molar mass, underpinned by precise atomic weights, highlights its importance in both theoretical and practical chemistry. Whether it’s for academic research or industrial application, the precision in molar mass calculations is non-negotiable for success in chemical endeavors.