Understanding the molecular geometry of N2 (nitrogen gas) is crucial for grasping fundamental principles in chemistry. While seemingly simple, this molecular configuration holds complex implications for physical properties, chemical reactivity, and molecular bonding. Here, we delve into practical insights to help you master the nuances of N2 molecular geometry through actionable steps, relatable examples, and problem-solving strategies.
Many students find the concept of molecular geometry daunting at first. It often appears as an abstract idea, but breaking it down into practical steps can make it understandable and even fascinating. Understanding N2 molecular geometry will help you in more advanced topics such as spectroscopy, chemical bonding, and even quantum mechanics.
Why N2 Molecular Geometry Matters
Knowing the molecular geometry of N2 is vital because it determines the molecule’s shape, which influences its physical properties and chemical behavior. Nitrogen gas, which makes up about 78% of Earth’s atmosphere, is a diatomic molecule with the formula N2. Its linear geometry may seem straightforward, but it provides a foundational building block for more complex molecules.
For chemists and engineers, understanding N2's geometry can help predict how nitrogen will interact with other molecules in different environments, whether in industrial applications or biological systems. Mastery of this concept enhances your ability to interpret data related to molecular interactions, chemical reactions, and even material properties.
Quick Reference
Quick Reference
- Immediate action item: Identify the bonding type in N2 (it is a triple covalent bond between two nitrogen atoms).
- Essential tip: Use the Molecule Shape Simulator tool to visualize the geometry of N2.
- Common mistake to avoid: Confusing N2 molecular geometry with that of a bent or tetrahedral molecule.
The Basics of N2 Geometry: A Detailed Overview
Let’s start with the basics. N2 is a diatomic molecule consisting of two nitrogen atoms bonded together. Each nitrogen atom has an atomic number of 7 and an electron configuration of 1s2 2s2 2p3. To understand the geometry of N2, it’s essential to delve into the concept of electron pairs and the valence shell electron pair repulsion (VSEPR) theory.
Electron Pairs and the VSEPR Theory
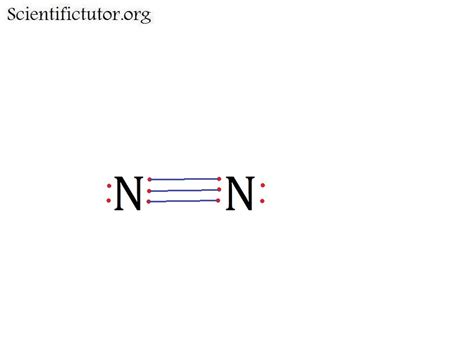
The VSEPR theory states that electron pairs around a central atom will arrange themselves to minimize repulsion. In N2, each nitrogen atom shares three electrons to form a triple bond, resulting in a linear shape. Each nitrogen atom provides three electrons to form three covalent bonds, and none of these bonds are considered “lone pairs” because they are all involved in bonding.
Step-by-Step Geometry Breakdown
Here’s a step-by-step guide to understanding N2 molecular geometry:
- Identify the number of atoms: N2 consists of two nitrogen atoms.
- Determine the bonding: Nitrogen atoms share three electrons each, forming a triple covalent bond.
- Apply the VSEPR theory: Since there are no lone pairs around the nitrogen atoms, the geometry will be linear. Each nitrogen atom pulls equally on the electrons, resulting in a 180-degree bond angle.
- Visualize the structure: Use molecular model kits or online simulation tools like the Molecule Shape Simulator to visualize the linear arrangement.
Advanced Aspects of N2 Geometry
Beyond basic understanding, deeper insights into the electronic structure and quantum mechanical aspects can enhance your comprehension.
Quantum Mechanical Explanation
N2’s molecular orbital theory offers a more advanced view. The molecule’s bonding is described by the overlap of atomic orbitals to form molecular orbitals. Specifically, the N2 molecule has a sigma (σ) bond formed by the head-on overlap of the 2s or 2p orbitals, along with two pi (π) bonds formed by the side-on overlap of the 2p orbitals.
Physical and Chemical Implications
The linear geometry of N2 means that its physical and chemical properties, like boiling and melting points, are influenced significantly. N2 is stable and unreactive under normal conditions, but it can participate in reactions under specific conditions, such as high temperatures or pressures.
Real-World Example: Industrial Applications
In industry, understanding N2’s molecular geometry helps in processes like nitrogen fixation in fertilizers or the use of nitrogen in controlled atmospheres for food preservation. The knowledge of its linear shape allows chemists to design reactors and processes that maximize the efficiency of these applications.
Practical Tips for Mastering N2 Geometry
Here are some practical tips to ensure you master N2 molecular geometry:
- Practice drawing the structure multiple times to internalize the linear arrangement.
- Use molecular model kits or online tools to physically manipulate the molecule and see how the atoms align.
- Solve practice problems that ask you to predict the geometries of different diatomic and polyatomic molecules.
- Review and reinforce your understanding by teaching the concept to someone else.
Common Misconceptions About N2 Geometry
It’s important to address some common misconceptions about N2 molecular geometry:
- Misconception: N2 is a bent molecule.
Clarification: N2 is linear due to the triple bond and absence of lone pairs, which causes the atoms to be aligned in a straight line.
- Misconception: N2 has only single bonds.
Clarification: N2 has a triple bond, which consists of one sigma (σ) bond and two pi (π) bonds.
- Misconception: The bond angle in N2 is different from 180 degrees.
Clarification: Due to its linear geometry, the bond angle in N2 is exactly 180 degrees.
FAQs on N2 Molecular Geometry
What is the bond angle in an N2 molecule?
The bond angle in N2 is 180 degrees. This is because N2 is a linear molecule, and the electron pairs or lone pairs around the nitrogen atoms arrange themselves to minimize repulsion. Thus, the nitrogen atoms are aligned in a straight line, creating a 180-degree bond angle.
How many electrons are involved in the bond between the nitrogen atoms in N2?
In an N2 molecule, each nitrogen atom contributes three electrons to form a triple bond. Therefore, a total of six electrons are involved in the bond between the nitrogen atoms.
Is N2 polar or nonpolar?
N2 is nonpolar. This is because it is a homonuclear diatomic molecule, meaning both nitrogen atoms have the same electronegativity. The symmetrical linear arrangement of the atoms means that any resulting dipole moments cancel each other out, resulting in a nonpolar molecule.
In summary, mastering N2 molecular geometry involves understanding its linear structure, the triple bond nature, and the physical and chemical implications of its shape. By following the practical steps outlined and using the real-world examples provided, you can gain a deep and practical understanding of N2 molecular geometry.