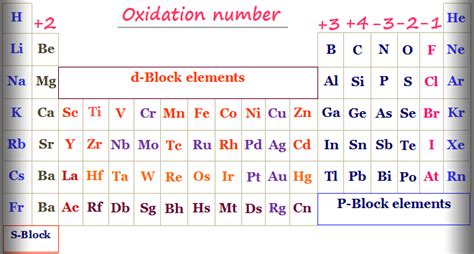
In the fascinating world of chemistry, oxidation numbers are pivotal to understanding reactions and their underlying mechanisms. If you've ever found the concept of oxidation numbers daunting or misunderstood it completely, this guide will reveal the hidden secrets to mastering them. We will delve into the specifics through actionable advice and real-world examples, addressing your pain points with clarity and precision. This guide is designed to not only demystify oxidation numbers but also to empower you with the practical skills you need to apply this knowledge confidently.
Understanding the Basics: What are Oxidation Numbers?
Oxidation numbers are assigned to atoms in a molecule to indicate the degree of oxidation (loss of electrons) of an atom in a chemical compound. They are fundamental in predicting the behavior of atoms in chemical reactions and help in balancing redox (reduction-oxidation) equations. Despite their importance, many find them confusing. Let’s start by breaking it down:
1. The oxidation number of an atom in its elemental form is zero. For example, in a single O2 molecule, each oxygen atom has an oxidation number of 0.
2. In a neutral molecule, the sum of oxidation numbers equals zero. For instance, in a water molecule (H2O), the oxidation number of hydrogen is +1, and oxygen is -2, making their sum 0.
3. In ions, the oxidation number is equal to the charge of the ion. For example, in a chloride ion (Cl-1), the oxidation number of chlorine is -1.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start with simple molecules like H2O and NaCl to practice assigning oxidation numbers.
- Essential tip with step-by-step guidance: Assign oxidation numbers by following these steps: (1) Assign oxidation numbers to all atoms in the molecule. (2) Check if the sum of oxidation numbers equals the total charge of the molecule or ion. (3) Ensure to update oxidation numbers for atoms when they form compounds.
- Common mistake to avoid with solution: Confusing oxidation numbers with actual charges. Remember, oxidation numbers reflect the hypothetical charge of an atom in a compound.
Detailed Guide: Assigning Oxidation Numbers Step by Step
Assigning oxidation numbers might seem intimidating at first, but by breaking down the process into manageable steps, it becomes straightforward:
Step 1: Identify the Element in Its Pure Form
When an element is in its pure form, its oxidation number is zero. For instance, in a piece of copper (Cu), each copper atom has an oxidation number of 0.
Step 2: Examine Neutral Molecules
In neutral molecules, the sum of the oxidation numbers is zero. Let’s use water (H2O) as an example:
Hydrogen typically has an oxidation number of +1, and since there are two hydrogen atoms, their combined oxidation number is +2. Oxygen typically has an oxidation number of -2, balancing the +2 from hydrogen to make the sum zero.
Step 3: Assign Oxidation Numbers in Polyatomic Ions
In polyatomic ions, the oxidation number total is equal to the charge of the ion. Take the nitrate ion (NO3-1) as an example:
Nitrogen in nitrate has an oxidation number of +5, and oxygen atoms each have an oxidation number of -2. The sum is +5 + 3(-2) = +5 - 6 = -1, matching the charge of the nitrate ion.
Step 4: Determine Oxidation Numbers in Complex Compounds
In more complex compounds like potassium permanganate (KMnO4), follow these sub-steps:
a.) Identify Elements with Known Oxidation Numbers:
Potassium (K) always has an oxidation number of +1, and in MnO4-1, oxygen always has an oxidation number of -2.
b.) Calculate the Total Contribution from Known Elements:
Four oxygen atoms each with an oxidation number of -2 contribute a total of -8, and one potassium with an oxidation number of +1 contributes +1.
c.) Solve for the Unknown:
The sum of oxidation numbers in KMnO4 must equal the overall charge of -1. Therefore, the oxidation number of manganese (Mn) must be +7 to balance the equation: +1 + (-8) + x = -1, solving for x gives us x = +7.
Advanced Techniques: Balancing Redox Reactions
Balancing redox reactions using oxidation numbers involves tracking the changes in oxidation states of individual atoms. Let’s break it down with an example:
Consider the redox reaction between zinc (Zn) and copper sulfate (CuSO4):
1. Identify oxidation numbers in the reactants: Zinc (Zn) in its elemental form has an oxidation number of 0, and copper (Cu) in CuSO4 has an oxidation number of +2.
2. Write the half-reactions:
Zinc half-reaction: Zn → Zn2+ + 2e- (oxidation)
Copper half-reaction: Cu2+ + 2e- → Cu (reduction)
3. Balance the half-reactions: The zinc half-reaction loses 2 electrons, so we need 2 electrons in the copper half-reaction. Multiply the copper half-reaction by 2 to balance electrons.
4. Combine and balance the overall reaction:
Zn + CuSO4 → ZnSO4 + Cu
Practical FAQ
Common user question about practical application
How do I know which element to assign first?
When assigning oxidation numbers, start with elements that have fixed oxidation states in most compounds. For example, hydrogen is usually +1, and oxygen is usually -2 in most cases. Additionally, for polyatomic ions, you can assign oxidation numbers starting with elements with known typical states, then solve for the remaining elements.
What should I do if the oxidation numbers don’t balance?
If the oxidation numbers don’t balance, double-check each assignment. Ensure you are assigning the correct oxidation number based on the typical states or known rules. Sometimes, re-assessing the bonds and charges in the molecule or ion will help identify where you went wrong.
Can oxidation numbers help in predicting reaction spontaneity?
Yes, oxidation numbers can help predict reaction spontaneity. Generally, redox reactions occur when there is a change in the oxidation states of elements involved. A spontaneous reaction typically involves a decrease in the overall oxidation state of the reactants. Understanding these changes helps predict the direction and likelihood of the reaction.
Through this guide, we've unlocked the secrets behind oxidation numbers, transformed complex ideas into simple actionable steps, and tackled some of the most common user pain points you might encounter. Remember, practice makes perfect. Utilize these tips and methods to master the subject confidently, applying these techniques in both theoretical and practical chemistry scenarios.