If you've ever wondered about the intricate dance of electrons within nitrogen trifluoride (NF3) and how it's represented through a Lewis structure, you're in the right place. Understanding the Lewis structure of this compound is pivotal in grasping its chemical behavior, reactivity, and potential applications. This guide will break down the process into simple, actionable steps, provide you with real-world examples, and address common pitfalls along the way. Let’s dive into the fascinating world of nitrogen trifluoride Lewis structure.
Understanding Your Need for Knowledge
Knowing the Lewis structure of NF3 is not just an academic exercise; it serves practical purposes in fields ranging from materials science to pharmaceuticals. If you’re here, it’s likely because you need to understand how to accurately represent this molecule or you’re curious about its electron distribution and how this impacts its reactivity. By breaking down the complexities of the NF3 Lewis structure, we aim to demystify this for you, so you can apply this knowledge effectively in your professional or academic endeavors.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start with the central atom nitrogen, as it will help you understand how to distribute valence electrons correctly.
- Essential tip with step-by-step guidance: Remember, nitrogen has 5 valence electrons, and each fluorine has 7. Properly distribute these electrons to satisfy the octet rule for each atom.
- Common mistake to avoid with solution: Avoid assuming that more electrons around fluorine will lead to stability. Stick to the correct number of electrons per atom to maintain a valid Lewis structure.
How to Draw the Lewis Structure for NF3
Drawing the Lewis structure for nitrogen trifluoride involves several straightforward steps. Follow these instructions to ensure you accurately depict this molecule.
Step 1: Identify the Total Number of Valence Electrons
To start, we need to tally the valence electrons of each atom involved. Nitrogen has 5 valence electrons, and since there are three fluorine atoms, each with 7 valence electrons, the total number of valence electrons is:
5 (from nitrogen) + 3*7 (from fluorines) = 5 + 21 = 26 valence electrons
This total will be used in constructing the Lewis structure.
Step 2: Choose the Central Atom
For NF3, nitrogen is the central atom because it is less electronegative compared to fluorine. Placing nitrogen in the center will ensure a balanced distribution of electrons.
Step 3: Connect the Central Atom to Each Outer Atom with Single Bonds
Now we use single bonds to link the nitrogen atom to each fluorine atom. This accounts for 6 electrons (2 electrons per bond, one from nitrogen and one from each fluorine).
Visually, this looks like:
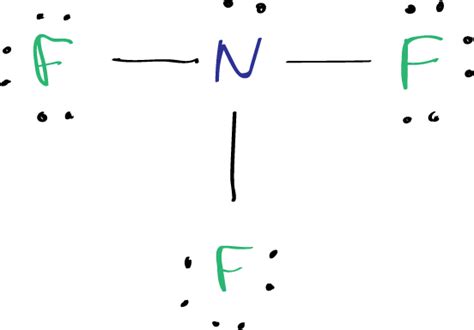
| F | — | N | — | F |
| | | | | | | | | |
| F |
Step 4: Distribute the Remaining Valence Electrons
We have 20 electrons left to distribute. According to the octet rule, each atom should aim to have 8 electrons around it.
Place 6 electrons (3 lone pairs) around nitrogen to complete its octet:
| F | — | [:N:] | — | F |
| | | | | | | | | |
| F |
Next, place 2 electrons on each of the three fluorine atoms to satisfy their octets:
| F | — | [:N:: | — | F |
| | | | | | | | | |
| [:F:] |
Step 5: Check the Octet Rule
Ensure all atoms fulfill the octet rule. Nitrogen has an expanded octet because it has 5 single electrons and 3 lone pairs, totaling 8 + 2 = 10 electrons around it. Fluorines each have 8 electrons around them.
| [:F:] | — | [:N:: | — | [:F:] |
| | | | | | | | | |
| [:F:] | (3 lone pairs) |
Step 6: Ensure Formal Charges are Minimized
Verify the formal charges on each atom. For nitrogen, the formal charge is zero (5 - 0.5*2 - 6 = 0). For each fluorine, the formal charge is also zero (7 - 0.5*6 - 2 = 0). The overall charge of the molecule is also zero.
Detailed How-To Section on Molecular Geometry and Electron Distribution
Understanding the geometry and electron distribution in NF3 helps in predicting its reactivity and interaction with other molecules. Here we delve deeper into the structural aspects and electron distribution.
Molecular Geometry
The nitrogen atom in NF3 is bonded to three fluorine atoms and has a lone pair of electrons. According to the VSEPR (Valence Shell Electron Pair Repulsion) theory, the shape of the molecule is determined by the repulsion between these electron pairs. For NF3, this results in a trigonal pyramidal geometry where the nitrogen atom is at the apex, and the three fluorine atoms form the base.
Electron Distribution
Nitrogen’s expanded octet (10 electrons around it) might seem unusual, but it’s perfectly valid according to contemporary chemical understanding, especially for second-row elements. The lone pair on nitrogen adds to the molecule’s polarity. Each bond between nitrogen and fluorine is a polar covalent bond, given the significant electronegativity difference between nitrogen and fluorine.
Reactivity and Interactions
NF3 is generally stable but can participate in reactions where it acts as a Lewis base (electron pair donor). Its polarity makes it reactive with electrophiles, allowing it to engage in various chemical processes. Understanding its Lewis structure aids in predicting these behaviors and planning reactions.
Practical FAQ
What are common mistakes people make when drawing the Lewis structure for NF3?
One common mistake is miscounting the total valence electrons. Ensure to add up all the valence electrons from nitrogen and the three fluorine atoms correctly. Another frequent error is not adhering to the octet rule, especially for nitrogen having an expanded octet. Always verify the formal charges to ensure they sum to zero for a neutral molecule.
How can understanding the Lewis structure of NF3 help in real-world applications?
Knowing the Lewis structure is fundamental in predicting the molecule’s behavior in chemical reactions. NF3 is