Understanding the nuances of stereochemistry is paramount for those delving into the intricate world of molecular structures. The R vs S configuration debate, particularly within organic chemistry, underscores the significance of chiral centers and the way they impact chemical reactions and biological functions. This discussion provides essential context for the advancement of pharmaceuticals and materials science, focusing on the fundamental principles that define molecular orientation.
Key Insights
- Understanding the R vs S configuration can significantly affect drug efficacy and safety.
- Chiral centers must be accurately identified to determine correct molecular behavior.
- Adopting rigorous stereochemical analysis techniques can lead to the development of better therapeutic compounds.
Organic compounds often possess chiral centers, which are carbon atoms bonded to four different groups. These centers lead to the formation of enantiomers—molecules that are mirror images of each other and non-superimposable, akin to left and right hands. The R vs S configuration system, proposed by Cahn, Ingold, and Prelog, is the universally accepted method for designating these chiral centers. It involves assigning priorities to the four substituents bonded to the chiral center and determining the direction of the priority sequence.
Fundamental Principles of R vs S Configuration
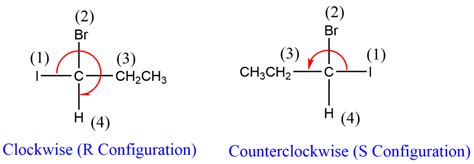
In this system, each group attached to the chiral center is assigned a priority based on the atomic number of its directly attached atom. The groups with higher atomic numbers get higher priority. Once priorities are established, the molecule is visualized so that the group with the lowest priority is oriented away from the observer. If the sequence from first to second to third priority groups is clockwise, the chiral center is designated as “R” (rectus, Latin for right). Conversely, if the sequence is counterclockwise, it is designated as “S” (sinister, Latin for left).This configuration is not merely an academic exercise; it holds practical implications. For instance, in pharmaceuticals, the two enantiomers of a chiral drug can exhibit vastly different effects. One enantiomer might be therapeutically beneficial, while the other could be harmful or completely ineffective. An example is thalidomide, where the R enantiomer has beneficial effects, whereas the S enantiomer caused severe birth defects.
Impact of R vs S Configuration in Drug Development
The impact of R vs S configuration in drug development cannot be overstated. The development of enantiopure drugs—those that contain only one enantiomer—can lead to more effective and safer therapies. Understanding stereochemistry allows chemists to design drugs with higher target specificity and reduced side effects. Additionally, accurate stereochemical analysis enables the creation of prodrugs, which are inactive compounds that are converted into active drugs in the body.Consider the example of the anti-migraine drug sumatriptan. Its development highlighted the importance of stereochemistry in drug efficacy. Through meticulous stereochemical analysis, the pharmaceutical company was able to optimize the active enantiomer, ensuring better therapeutic outcomes and minimizing risks.
How does R vs S configuration affect biological activity?
The R vs S configuration can lead to drastically different biological activities of enantiomers. The different spatial orientations can result in varying interactions with biological targets such as enzymes and receptors, leading to different therapeutic effects or even toxicity.
What techniques are used for determining R vs S configuration?
Several techniques are used, including nuclear magnetic resonance (NMR) spectroscopy, optical rotation measurements, and X-ray crystallography. These methods allow chemists to determine the spatial arrangement of atoms and assign the appropriate R or S configuration.
The R vs S configuration debate is more than just an academic discussion; it plays a crucial role in various scientific fields, particularly in chemistry and pharmacology. Through precise determination and understanding of chiral centers, significant advancements in drug development and molecular science can be achieved, ultimately contributing to better health outcomes and innovative materials.