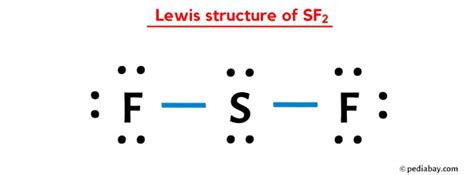
If you’ve ever found yourself tangled in the complexity of deciphering the SF2 Lewis structure, you’re not alone. Understanding the layout and reasoning behind this specific molecule’s structure is crucial for mastering the fundamentals of chemical bonding and molecular geometry. This guide will walk you through the intricacies of the SF2 Lewis structure, offering clear and practical insights that will illuminate even the most confusing aspects.
Understanding SF2 Lewis Structure: A Step-by-Step Guide
The SF2 molecule, comprising one sulfur (S) atom and two fluorine (F) atoms, may seem straightforward at first glance, but the real challenge lies in accurately representing its electron configuration. This guide breaks down the process into digestible steps, ensuring you can grasp the core principles and apply them effectively.
Why SF2 Lewis Structure Matters
The SF2 Lewis structure is a crucial concept in understanding molecular geometry, polarity, and reactivity. It provides a visual representation of how atoms in a molecule are bonded and the spatial arrangement they form, which is vital for predicting chemical behavior and interactions.
Getting Started: Problem-Solution Opening
Deciphering the SF2 Lewis structure can be daunting. The initial steps involve identifying the total number of valence electrons, determining the central atom, and figuring out how to place the electrons to satisfy the octet rule for all atoms involved. Missteps here can lead to incorrect molecular shapes and faulty predictions about the molecule’s properties. This guide aims to demystify these steps, offering clear, actionable advice to ensure you arrive at the correct SF2 Lewis structure.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start by counting the total valence electrons for both sulfur and fluorine atoms.
- Essential tip with step-by-step guidance: Place the least electronegative atom in the center to form the backbone of the structure.
- Common mistake to avoid with solution: Forgetting to distribute lone pairs and ensure all atoms achieve an octet.
How to Determine the SF2 Lewis Structure
Let’s dive into the detailed process of determining the SF2 Lewis structure, ensuring you grasp each step for a comprehensive understanding.
Step 1: Count Valence Electrons
The first step in drawing the SF2 Lewis structure is to determine the total number of valence electrons. Sulfur (S) is in group 16 of the periodic table, so it has six valence electrons. Each fluorine (F) atom, located in group 17, has seven valence electrons. Since there are two fluorine atoms, you multiply this by two. Therefore, the total number of valence electrons available for the SF2 molecule is:
6 (from sulfur) + 7*2 (from two fluorines) = 20 valence electrons
Step 2: Identify the Central Atom
In the SF2 molecule, sulfur is less electronegative than fluorine. Therefore, sulfur will be the central atom. This is a key decision point because the placement of atoms affects the structure’s overall geometry.
Step 3: Connect the Central Atom to Surrounding Atoms
Place the sulfur atom in the center and connect it to each of the two fluorine atoms by drawing single bonds. Each bond consists of two electrons, which accounts for four electrons right away. Since there are two fluorine atoms, you’ll need two bonds.
Step 4: Distribute Remaining Electrons
After forming bonds, distribute the remaining electrons to achieve an octet for all atoms. First, complete the fluorines’ octets. Each fluorine atom will have three lone pairs (six electrons) to satisfy its octet rule. Next, place any remaining electrons on the sulfur atom.
Let’s break it down: You started with 20 valence electrons and have already used 4 for the two single bonds. Now, subtract those from the total:
20 - 4 = 16 electrons remaining
Distribute these electrons starting with the fluorines:
Each fluorine already has six electrons (three lone pairs). Distribute the remaining 16 electrons (8 pairs) as lone pairs on the fluorines and then on sulfur. This ensures each atom has an octet.
Step 5: Check the Octet Rule and Resonance
Ensure all atoms have a complete octet. Sulfur, however, might not end up with eight electrons because it’s often more stable with a lower electron count in hypervalent molecules. In SF2, sulfur forms two single bonds with two fluorines and has no lone pairs, giving it only four electrons. This is perfectly fine, as sulfur can expand its octet beyond the traditional octet rule.
Resonance structures exist where the electrons could be delocalized differently, but for SF2, this direct arrangement is the most stable.
Advanced Insights: Optimizing SF2 Lewis Structure Accuracy
For a molecule like SF2, precision is key. As you become more comfortable with these structures, consider advanced nuances such as electronegativity differences influencing bond types and geometries, and how hypervalency affects central atom stability.
Practical FAQ
What happens if I place fluorine in the center?
If you place fluorine in the center, it wouldn’t be correct because fluorine is more electronegative and would not typically act as the central atom in a molecule. It would also lead to an unstable structure since fluorine atoms, being highly electronegative, would pull electrons away from the less electronegative atoms, leading to an imbalance in the molecule’s overall structure and properties.
How do I know if my SF2 Lewis structure is correct?
A correct SF2 Lewis structure will have sulfur at the center with two single bonds to two fluorine atoms and no lone pairs on sulfur. Each fluorine should have three lone pairs to fulfill the octet rule. Ensure you’ve used all 20 valence electrons correctly and the structure is stable.
Final Tips for Mastery
Mastering the SF2 Lewis structure is a gateway to understanding more complex molecules. Remember, practice is key. Drawing multiple Lewis structures will solidify your understanding, so don’t hesitate to explore other molecules to broaden your skills further.
Use these insights to delve deeper into molecular chemistry, and remember, every expert was once a beginner at the start of their journey. With dedication and practice, you’ll unlock the mysteries of molecular structures one step at a time.