Are you curious about the SO3 compound? You're not alone. Understanding chemical compounds like SO3 can seem daunting at first, but once you break it down, it becomes quite fascinating. In this guide, we will dive deep into the SO3 compound name, offering clear, actionable advice to demystify this common yet complex chemical. By the end of this, you'll not only know what SO3 stands for but also how to apply this knowledge in real-world scenarios. Let’s begin our journey to unlock the mystery of SO3!
Introduction: The Challenge and Importance of Understanding SO3
SO3, or sulfur trioxide, is an important chemical compound that features prominently in both industrial and academic contexts. Despite its intimidating name, the essence of SO3 can be understood with a few clear steps. For anyone who needs to work with or understand chemicals in fields such as chemistry, environmental science, or engineering, grasping the basics of SO3 is crucial. The compound’s role in forming sulfuric acid, a key industrial chemical, highlights its significance.
This guide aims to address the common user pain points when first encountering SO3. We will lay out the problem—understanding and utilizing SO3 effectively—and then provide a solution through step-by-step guidance, real-world examples, and practical tips.
Quick Reference
- Immediate action item: Familiarize yourself with the chemical formula SO3 and its composition—one sulfur atom and three oxygen atoms.
- Essential tip: To comprehend SO3 better, consider its role in sulfuric acid production.
- Common mistake to avoid: Confusing SO3 with other sulfur oxides, like SO2. Ensure you understand the difference in their molecular structures.
Breaking Down SO3: From Basics to Advanced Understanding
To understand SO3 fully, we’ll start with its fundamental elements and move toward its practical applications.
Basic Structure of SO3
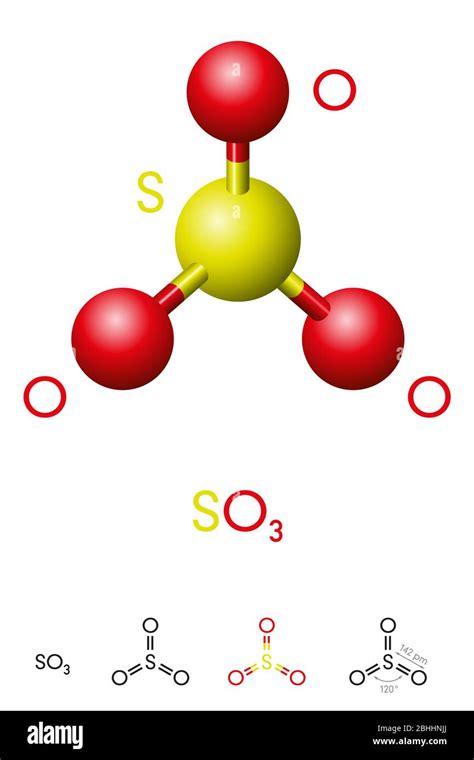
SO3 is composed of one sulfur (S) atom bonded to three oxygen (O) atoms. Understanding this structure is foundational to grasping its behavior and uses.
Here’s a simple way to visualize it:
- Sulfur atom at the center
- Three oxygen atoms surrounding the sulfur, each forming a double bond
This structural setup gives SO3 its unique properties, which we will explore in more detail.
Molecular Geometry and Bond Angles
The SO3 molecule is trigonal planar, meaning it has a flat, triangle-like structure with bond angles of 120 degrees. This geometry is a direct result of the double bonds formed between the sulfur and each oxygen atom.
Visualizing this geometry helps in understanding its chemical behavior, such as its reactivity in forming sulfuric acid.
Electronegativity and Polarity
Sulfur and oxygen have different electronegativities, which leads to polar covalent bonds within the SO3 molecule. Each sulfur-oxygen bond is polar, with oxygen being more electronegative than sulfur.
Here’s how it works:
- Oxygen atoms attract electrons more strongly, making each bond slightly negative.
- The sulfur atom remains slightly positive.
This polarity is vital when considering how SO3 interacts with other compounds, especially in industrial applications.
How SO3 Forms and Converts into Other Compounds
Understanding the transformation of SO3 into other compounds, particularly sulfuric acid, is key to its practical application.
Formation of SO3 from SO2
Sulfur dioxide (SO2) can be oxidized to form SO3, a critical step in industrial processes such as acid rain prevention and sulfuric acid production.
The chemical reaction can be represented as:
2SO2 + O2 → 2SO3
This reaction typically requires a catalyst, such as vanadium(V) oxide (V2O5), to increase the reaction rate.
Conversion of SO3 to Sulfuric Acid (H2SO4)
SO3 is a crucial intermediate in the production of sulfuric acid. The final step involves combining SO3 with water:
SO3 + H2O → H2SO4
This step is typically conducted in two stages:
- First, SO3 is absorbed into concentrated sulfuric acid, forming oleum (H2S2O7).
- Then, water is added to the oleum to form sulfuric acid:
- H2S2O7 + H2O → 2H2SO4
This method maximizes efficiency and avoids the explosive reaction that can occur if water is added directly to SO3.
Practical Applications of SO3
Knowing how to practically apply SO3 in real-world scenarios is the ultimate goal of this guide.
Industrial Uses of SO3
SO3 is extensively used in industries to manufacture sulfuric acid, which is a fundamental chemical for producing fertilizers, detergents, dyes, and numerous other compounds.
Here’s how it plays a role in these processes:
- Fertilizers: Sulfuric acid is used to produce ammonium sulfate, a common fertilizer.
- Detergents: It is a key ingredient in the production of laundry detergents.
- Dyes: Sulfuric acid is used in the synthesis of various dye compounds.
Environmental Considerations
While SO3 has numerous industrial benefits, it’s important to consider its environmental impact.
When released into the atmosphere, SO3 can contribute to acid rain, a harmful environmental pollutant.
Here’s how industries mitigate these effects:
- Installing scrubbers to remove SO3 emissions.
- Implementing catalytic converters to oxidize SO2 to SO3 more efficiently.
Safety Measures when Handling SO3
Handling SO3 requires careful attention due to its corrosive and reactive nature.
Consider these safety measures:
- Use appropriate personal protective equipment (PPE) such as gloves and goggles.
- Store SO3 in well-ventilated areas away from incompatible materials.
- Ensure proper training for workers handling SO3 to prevent accidents.
Following these guidelines helps to maintain a safe working environment.
Can SO3 react directly with water to form sulfuric acid?
No, SO3 should not react directly with water due to the highly exothermic nature of the reaction, which can be dangerous. Instead, it is safely converted to sulfuric acid through a controlled process involving oleum (H2S2O7).
Advanced Techniques for Using SO3
For those looking to dive deeper into the applications of SO3, here are some advanced techniques:
Catalytic Conversion Optimization
Improving the catalytic conversion of SO2 to SO3 can significantly enhance industrial efficiency:
- Optimizing the catalyst type (e.g., using more advanced formulations of vanadium oxide).
- Operating the reactor at optimal temperatures and pressures to maximize conversion rates.
- Regularly maintaining and replacing catalysts to ensure their efficiency over time.
Recycling and Reuse of By-products
Efficient industrial processes involve the recycling and reuse of by-products to minimize waste and environmental impact:
- Recycling waste sulfuric acid back into production to reduce raw material use.
- Utilizing by-products in secondary processes, such as using any unreacted SO2 in other chemical syntheses.
This guide has taken you from the basics of understanding the SO3 compound to advanced industrial applications. With this knowledge, you’re now equipped to not only grasp the significance of SO3 but also to utilize