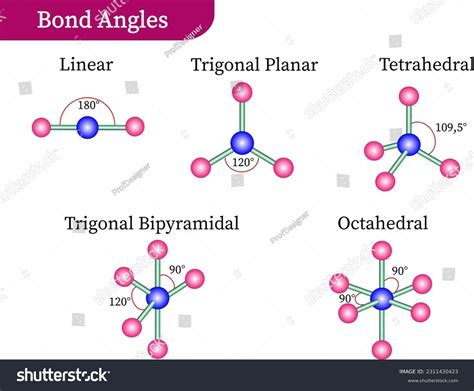
Understanding the intricacies of trigonal bipyramidal molecular geometry can revolutionize the way chemists approach molecular interactions and chemical reactions. This unique 3D configuration, defined by its five-atom framework, is crucial in fields ranging from inorganic chemistry to materials science. The symmetrical balance and spatial arrangements inherent in this structure offer valuable insights into molecular behavior and bonding.
Primary Insight: The Symmetry and Stability of Trigonal Bipyramidal Structures
The trigonal bipyramidal geometry is an arrangement where five atoms are situated in a central atom. Three atoms lie in a plane with 120-degree angles, forming a trigonal base, while two atoms occupy positions above and below this plane, axially positioned with the central atom. This configuration offers an ideal understanding of the balance and stability critical for various molecular interactions. Its symmetrical design minimizes repulsive forces between atoms, contributing to a robust, stable molecular framework.
Key Insights
- Primary insight with practical relevance: The symmetrical and balanced nature of trigonal bipyramidal structures enhances the stability of molecules.
- Technical consideration with clear application: This geometry is crucial in explaining the reactivity and orientation of certain molecules in chemical reactions.
- Actionable recommendation: Leveraging this understanding can aid in designing more effective catalysts and materials in chemical engineering.
Application in Chemical Reactions
The trigonal bipyramidal geometry plays a pivotal role in the realm of chemical reactions, particularly during the transition states of various processes. For example, in the SN2 reaction, the formation of a transition state often adopts this geometry, facilitating the nucleophilic attack and leaving group departure in a smooth, balanced manner. This understanding can greatly benefit chemists when predicting reaction mechanisms and designing new synthetic pathways.
Geometrical Implications in Coordination Chemistry
In coordination chemistry, trigonal bipyramidal geometry often dictates the spatial orientation of ligands around a central metal atom. A prominent example is found in certain transition metal complexes, where five ligand sites align in this geometry to maximize their individual interactions while minimizing steric hindrance. This understanding helps chemists in designing metal-organic frameworks (MOFs) and catalysts that are efficient and robust under various operational conditions.
What is an example of a molecule with trigonal bipyramidal geometry?
One classic example is the molecule PCl5, where phosphorus is surrounded by five chlorine atoms forming a trigonal bipyramidal shape.
Why is the trigonal bipyramidal geometry less common than other geometries?
This geometry is less common due to the increased steric strain between axial atoms compared to equatorial atoms, making it less stable in many scenarios.
The exploration of trigonal bipyramidal molecular geometry unveils the nuanced intricacies of molecular stability and reactivity. This knowledge is instrumental for advanced research and practical applications in chemistry and materials science, promising more effective, innovative solutions to complex chemical problems. By integrating this understanding, professionals can pave the way for groundbreaking advancements in molecular design and chemical engineering.