Understanding the Van der Waals Equation: A Precise Guide for Real Gases
In the realm of physical chemistry, comprehending the behavior of gases goes beyond the scope of the Ideal Gas Law. For real gases, the Ideal Gas Law often falls short due to the significant volume of gas molecules and intermolecular forces, making the Van der Waals equation a more accurate tool. Here, we will explore the nuances of the Van der Waals equation, offering practical insights and applications for real gas behavior analysis.
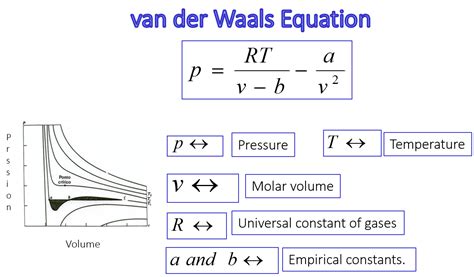
The Van der Waals equation is a refined model tailored to account for the actual volume of gas molecules and intermolecular attractions. It modifies the Ideal Gas Law by incorporating two constants: one that adjusts for the volume of gas particles (a), and another that corrects for the attractive forces between particles (b).
Key Insights
- Primary insight with practical relevance: The Van der Waals equation provides more accurate predictions of gas behavior under non-ideal conditions.
- Technical consideration with clear application: By reducing the volume occupied by gas particles and accounting for intermolecular attractions, this equation helps predict gas behavior under a wide range of pressures and temperatures.
- Actionable recommendation: Utilize the Van der Waals equation for analyzing gases in industrial applications and research where high accuracy is required.
Real Gas Behavior and the Van der Waals Equation
In contrast to the Ideal Gas Law, which presumes particles occupy no volume and exhibit no intermolecular forces, the Van der Waals equation addresses these assumptions. The equation is given as:
(P + a(n/V)^2)(V-nb) = nRT
Here, P represents pressure, V is the volume, n is the number of moles, T is the temperature, R is the universal gas constant, and a and b are Van der Waals constants specific to each gas. The 'a' constant modifies the pressure term, accounting for the attractive forces that pull molecules together, thereby reducing the pressure exerted. Meanwhile, the 'b' constant adjusts the volume term, subtracting the volume occupied by the gas molecules themselves, thus increasing the available volume.
Comparative Analysis of Ideal and Van der Waals Models
While the Ideal Gas Law provides a foundational understanding of gas behavior, it often does not suffice for gases under high pressures or low temperatures, where deviations from ideal behavior become pronounced. The Van der Waals equation’s modifications to both the pressure and volume terms enable it to capture these deviations more accurately. For instance, the equation’s applicability in regions where the Ideal Gas Law fails is vital for industries that deal with liquefied gases and high-pressure systems.
Consider a practical example involving hydrogen gas: under high-pressure conditions, the ideal assumption that molecules do not occupy space breaks down, causing significant deviations from predicted behavior. Utilizing the Van der Waals equation, hydrogen’s behavior can be modeled more precisely, ensuring more accurate predictions and safer industrial practices. The constants a and b for hydrogen allow for better alignment with experimental data, demonstrating the equation's practical utility.
What are the primary differences between the Ideal Gas Law and the Van der Waals equation?
The Ideal Gas Law assumes that gas particles occupy no volume and exhibit no intermolecular forces, which simplifies calculations but often fails under non-ideal conditions. The Van der Waals equation corrects for the volume of gas particles and the intermolecular forces, offering a more accurate model for real gases under a wider range of conditions.
How do the Van der Waals constants (a and b) influence gas behavior?
The constant 'a' adjusts for the attractive forces between gas molecules, reducing the calculated pressure by acknowledging that molecules attract each other, thus reducing the pressure exerted on container walls. The constant 'b' subtracts the volume occupied by gas molecules from the total volume, reflecting the fact that gas molecules have space that must be accounted for, thereby increasing the effective volume.
In conclusion, the Van der Waals equation represents a significant advancement in the field of gas laws, offering enhanced accuracy for real gas behavior analysis under non-ideal conditions. Through a clear understanding and application of this refined model, chemists and engineers can ensure precise predictions, benefiting various industrial applications and research fields.