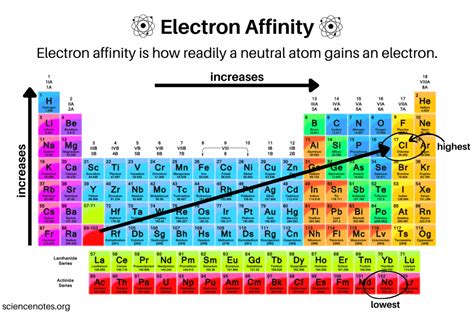
Understanding electron affinity is pivotal for both theoretical and applied chemistry. At its core, electron affinity is the amount of energy released when an isolated gaseous atom or molecule acquires an additional electron to form a negative ion. This fundamental property influences chemical reactivity, molecular stability, and the behavior of substances in various environments.
Key insights:
Key Insights
- Electron affinity is crucial for predicting how elements will behave in chemical reactions.
- It helps in understanding the stability of anions in different chemical compounds.
- High electron affinity values suggest a stronger likelihood for elements to form anions.
To comprehend electron affinity in practical terms, consider its significance in biochemical reactions. The process of ATP synthesis in cellular respiration relies on the electron affinity of specific molecules, particularly in the redox reactions that convert ADP into ATP.
One cannot overstate the importance of electron affinity in determining the thermodynamic feasibility of various chemical processes. A deep dive into the periodic table reveals that elements in Group 17 (halogens) have high electron affinity values, making them highly reactive. Chlorine, for instance, has a high electron affinity, which explains its readiness to form chloride ions (Cl-) in reactions.
The second area of interest lies in the realm of material science, where electron affinity plays a critical role in designing semiconductors and photovoltaic materials. Silicon, a quintessential semiconductor, has a well-known electron affinity that influences its electrical conductivity and its ability to trap or release electrons under varying conditions.
Chemical Behavior of Elements
The chemical behavior of elements can be extensively studied through electron affinity. This property not only determines the reactivity of an element but also sheds light on its potential to form ionic compounds. For instance, alkali metals in Group 1 (such as sodium and potassium) have relatively low electron affinities compared to non-metals in Group 17. This is because non-metals have smaller atomic radii and higher effective nuclear charges, allowing them to attract an additional electron more energetically favorable.Elements with high electron affinity values tend to form anions readily. This is crucial in understanding various chemical and biochemical processes where anions play a vital role. For instance, in biological systems, the high electron affinity of oxygen allows it to readily form O2- ions, playing a fundamental role in respiration.
Applications in Material Science
Electron affinity is not merely a theoretical construct; it has profound practical applications in material science. Semiconductors, which form the backbone of modern electronic devices, are deeply influenced by the electron affinity of constituent materials. For instance, gallium arsenide (GaAs) is widely used in high-speed electronics and optoelectronic devices due to its distinct electron affinity, enabling efficient electron movement and thus superior device performance.Additionally, photovoltaic materials such as cadmium telluride (CdTe) rely on precise electron affinity values to optimize the generation of electrical power from sunlight. These materials are engineered to efficiently absorb light and convert it into electron flow, which can then be harnessed to generate electricity.
How does electron affinity affect chemical reactions?
Electron affinity impacts the likelihood of an atom gaining an electron to form a negative ion. Elements with high electron affinity are more likely to participate in reactions where they gain an electron, thus influencing the overall chemical behavior and reactivity of the substances involved.
Why is electron affinity important in the development of semiconductors?
In semiconductor materials, electron affinity determines how easily electrons can be added or removed, which in turn affects the material's conductivity and its efficiency in electronic devices. This property is crucial for designing devices with desired electrical characteristics.
In conclusion, electron affinity is a cornerstone concept in chemistry, influencing both the fundamental understanding of chemical reactions and the advanced applications in material science. Through grasping its significance and implications, we can better predict and manipulate the chemical and physical properties of various elements and compounds.